|
In this activity, students review ideas connected with the structure of atoms using a set of ‘similar and different’ cards, featuring key phrases relevant to the topic. RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation.Good insulators, conductors, and semi-conductors are usually refined materials. Silicon is the most common semi-conductor. Semi-conductors can conduct electricity at a certain level without being damaged however, semi-conductors can be damaged with too much electric force. The atoms of a semi-conductor have no free electrons however, they do have an electron that is bonded rather loosely and can be shared with the application of a moderate electric force. Semi-conductors are insulators that lack commitment. Materials such as rubber and glass are good insulators. 
The flow of electrons in an insulator is relatively uncommon, and when it does occur it tends to be accompanied by intense heat, loud noise, melting or explosions, smoke and/or flame. They have no free electrons, and it takes a great deal of electric force to get the electrons of an insulator to flow. Insulators are quite selfish about sharing their electrons. Metals such as copper, silver, and gold are good conductors. With very little force, they can be traded to other atoms, or flow to create electricity. "Free electrons" are electrons that are very weakly bonded to the atom. This is due to electrons called "free electrons", which exist in the atoms of conductors. Very little electric force is required to cause electron flow in conductors. We can classify matter into three categories with regard to electron flow, conductors, insulators, and semi-conductors.Ĭonductors are materials that conduct electricity easily. To conclude the discussion of atomic structure, let us consider the materials in which electrons flow. The balance between force and resistance will be quantified in the section on electric circuits. When the balance tips, either with greater electric forces, or with lower resistance to electron flow, electricity is created. In order for electrons to flow, the electric forces must overcome the resistance to electron flow. This is because a balance between electric forces, and resistance to electron flow, are maintained in matter most of the time. So, all matter has electrons, ready to flow, yet electricity is not randomly buzzing everywhere, zapping stuff. This flow of electrons is what we call electricity. In addition, large numbers of adjacent atoms can share electrons in a long chain, from one atom to the next to the next, and so forth, creating a flow of electrons. Atoms will share electrons with other atoms to form bonds, creating molecules and compounds, solids and gasses - in short, all matter. Atoms typically like to keep a neutral charge, so if they borrow an electron from one atom, they tend to share one out with another atom. Helium, for instance, has two protons and two electrons.Īn atom will keep its protons and neutrons to itself.  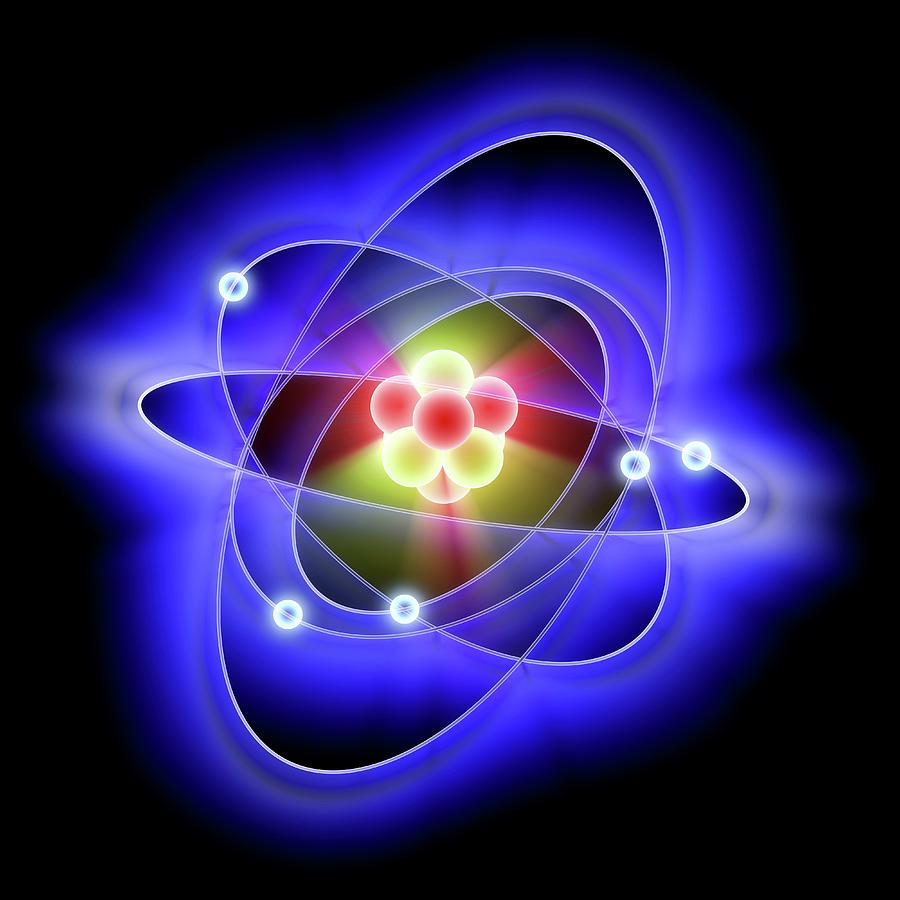
The number of protons in an atom determines what kind of element the atom is, and it is expected that an atom will have the same number of electrons as protons in order to balance the atom's charge. All other elements have the same basic structure: protons and neutrons at the center, and electrons orbiting around. 
Similarly, a heavy proton sits at the center of the hydrogen atom, while the electron orbits the proton with great speed. In the center is the large heavy earth, and around it orbits the smaller moon. The structure of a hydrogen atom can be thought of as like the earth and its moon. Hydrogen is the simplest of the elements a hydrogen atom consists of one proton and one electron. Electrons are negatively charged particles, and are relatively light compared to protons and neutrons. Protons are positively charged particles, neutrons are uncharged particles, and both are heavy in the subatomic scheme of things. Atoms of all elements are made up of three basic building blocks, protons, neutrons, and electrons. Most people are familiar with the names of elements such as oxygen, nitrogen, iron, gold, and hydrogen. The different kinds of atoms are called elements, and are listed in a pictorial display called the periodic table of elements. All matter consists of tiny pieces of stuff called atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
